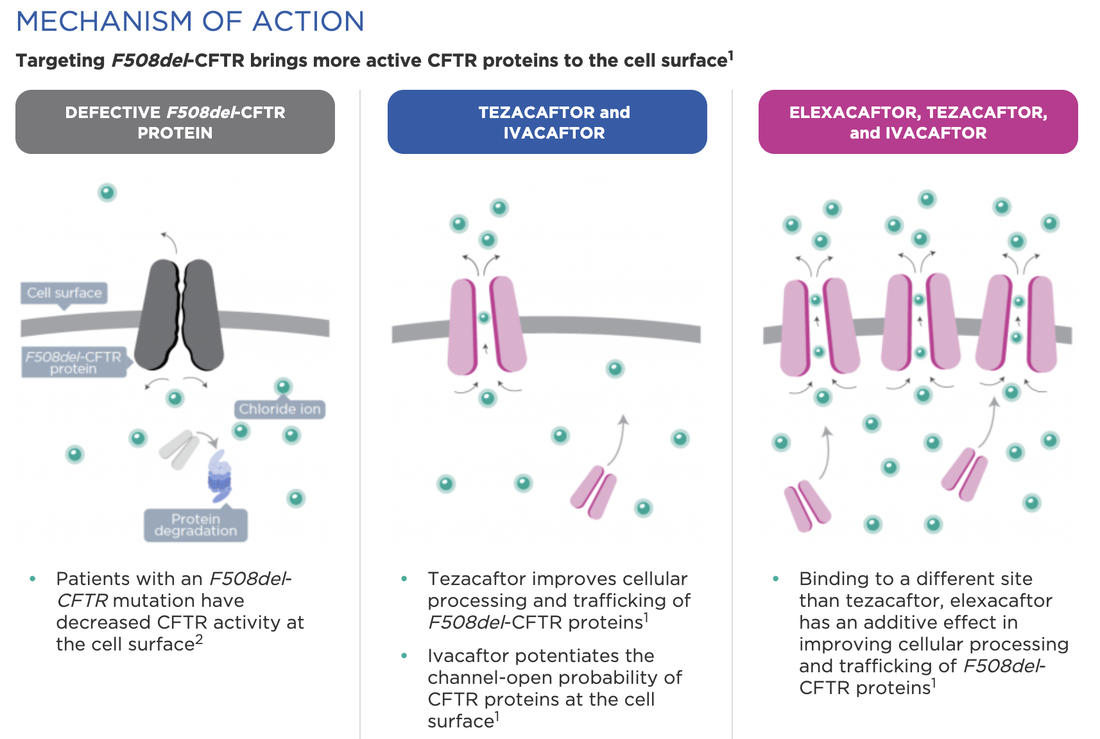
By Samitha NemirajaiahCystic fibrosis is a progressive, debilitating monogenic genetic disorder, affecting 1 in 3000 births. Cystic fibrosis is caused by a mutation in the CFTR protein (Cystic Fibrosis Transmembrane Conductance Regulator). Cystic fibrosis has no cure, but in the last decade, there has been a breakthrough in drug development by Vertex pharmaceuticals, resulting in the drug Trikafta. Trikafta is not a gene replacement therapy, but it does target the gene mutations, unlike the current medications that just treat the symptoms. Last year, the FDA (The Food and Drug Administration) approved this breakthrough therapy Trikafta, developed by Vertex Pharmaceuticals., which is a combination of three drugs: Tezacaftor, Ivacaftor, and Elexacaftor, all of which target the defective CFTR protein. The efficacy of Trikafta compared to current therapies is superior based on clinical trials. Over the last eight years, the FDA has approved these drugs individually and, eventually, a combination covering 90% of cystic fibrosis patients. Trikafta clinical trials demonstrated a very robust improvement in lung function, allowing the FDA to fast-track the approval based on robust results. There have been nine FDA approvals and 105 clinical trials conducted for these three drugs individually or in combination. Based on these results. these drugs have improved the mortality rate and quality of life for cystic fibrosis patients. In its simplest form, cystic fibrosis can be explained as a decreased activity of cellular processing at the cell surface. When there is a mutation or defect in a gene, then it cannot produce the required proper protein and, in this case, resulting in a defective CFTR protein. The defective CFTR protein decreases the flow of chloride ions through channels at the epithelial surfaces of the lung, sinuses, and pancreas, causing mucus to build-up resulting in respiratory issues. Trikafta contains two correctors and one potentiator of the CFTR channel. Ivacaftor works as a potentiator by opening the channels or gates, thereby increasing the flow of chloride ion, whereas Tezacaftor works as a corrector improving the CFTR function. Elexacaftor, which is another corrector, further enhances the functionality of the CFTR protein. The drug helps to transport the chloride ions and opens the channels for the movement and reducing the obstruction. According to pediatric pulmonologist Dr. Nathan Kraynack M.D., who directs The Cleveland Clinic’s children cystic fibrosis program, the triple combo drug Trikafta is a game-changer. He said, “I don’t think it’s quite a cure, but it significantly improves patient lung function. It can reduce symptoms and dramatically improve quality of life, and it is something that no one really could foresee 20 to 25 years ago.” He also said, “There is a reason for optimism.” He further states that “We have come so far in just the last ten years that I foresee a cure during many of my patients’ lifetimes” (Triplet CFTR Modulators 2018) Currently, there are many Gene replacement therapy-based clinical trials underway to find a cure for cystic fibrosis. References:
0 Comments
Leave a Reply. |
ABOUTSubmit your work to be considered for publication in the Newly Created SNHS Newspaper! CategoriesOuter Space History Biology
|