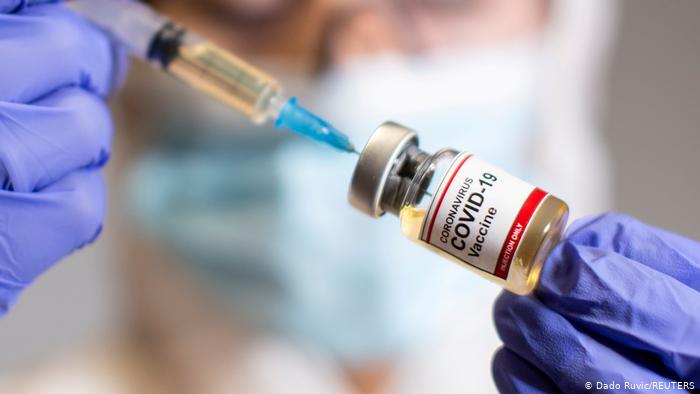
BY Anya GandavadiAs of March 29, all adults in Texas will be eligible to receive a COVID-19 vaccine, almost exactly one year after the first lockdown was initiated in the United States of America. “Before the COVID-19 pandemic, getting a new vaccine from concept to approval could take 10 years and billions of dollars.” So how was the COVID-19 vaccine developed so quickly? For two main reasons. 1. Much of the red tape has been cut away, and 2. There has been worldwide collaboration unlike never before. Pharmaceutical companies have been “getting a boost from Operation Warp Speed, a collaboration between the pharmaceutical industry and the federal government. To offset the cost of the development of the COVID-19 vaccine and to help mobilize approved vaccines as quickly as possible to the American public, the government established nearly $10 billion in federal funds. This has greatly accelerated the timeline for the development of vaccines through clinical trials, FDA review and mass distribution of a vaccine.” With a combination of existing vaccine technology and newer mRNA technology, and with scientists from all over the world working on the vaccine, one can be assured that these vaccines are safe and valuable although they were developed rapidly. So far in the US, over 147 million doses have been administered and over 16% of America’s population is fully vaccinated. Through continuing trials, scientists have been able to gather reliable data on the side effects of the 3 major COVID-19 vaccines. According to the CDC, one can expect more side effects from the second booster dose of the vaccine as your body is already prepared with the antibodies to attack the COVID-19 virus spike. An exeption is the Johnson & Johnson Vaccine which has only one dose and minimally lower protection rates, and recipients typically experience similar symptoms. The most common of these side effects is injection site pain or swelling which was experienced by nearly 92% of participants in Moderna's clinical trial, 84% in Pfizer’s trial, and 49% in Johnson & Johnson's. “Other common side effects include fatigue, headache, and body or muscle aches. About 65% of vaccine recipients in Pfizer's and Moderna's trials, and 38% in Johnson & Johnson's, developed fatigue.” A small study also found that for those who haven't had COVID-19 before, side effects tended to be more numerous and severe after the second dose. This is likely because someone who has had the COVID-19 infection would be able to develop a quicker and more robust reaction to being re-exposed. Additionally, “roughly twice as many participants in Pfizer's trial developed chills and joint pain after their second dose than after their first. In Moderna's trial, meanwhile, about five times as many participants developed chills after their second dose as did after their first. Fevers were also far more common among second-dose recipients than first-dose recipients in both trials.” The vaccine also affects different demographics of people differently. “The Centers for Disease Control and Prevention analyzed Americans' reactions to nearly 14 million doses of the Pfizer and Moderna shots from December to January. The results showed that roughly 79% of instances of vaccine side effects reported to the CDC came from women, though just 61% of doses were administered to women overall.” Scientists suspect that this has to do with the presence of the hormone estrogen which is an immune stimulant, versus testosterone which is an immune suppressant. Additionally, immune systems deteriorate with age, so younger people can expect to have more sever symptoms than older people. Regardless, severe and life threatening reactions to the COVID-19 vaccine, like other vaccines are extremely rare, mostly coming from a handful of cases of anaphylaxis. It is important that people take the COVID-19 vaccine to begin developing herd immunity to protect those who are in danger of dying. “People with weakened immune systems don't mount a strong defense against viral infections in general, so they're particularly vulnerable to severe COVID-19. For that reason, the CDC recommends that these groups get vaccinated right away.” As Kathleen Mullane, the director of infectious disease clinical trials ar the University of Chicago Medicine, says, “Even the most effective vaccine can’t protect us or our loved ones if people are afraid to take it or will not take it.” References:
0 Comments
Leave a Reply. |
ABOUTSubmit your work to be considered for publication in the Newly Created SNHS Newspaper! CategoriesOuter Space History Biology
|